|
The Modern Periodic Table's 118 elements are mentioned below-Īlso, read about- Father of Chemistry and the Difference Between Soap and Detergent. Read more about Chemistry Periodic Table and the First 20 Elements in the Periodic Table. Furthermore, several of the "fudged" attributes were later recalculated and found to be substantially closer to his predictions. There were many skeptics, and it took years for Mendeleev's patterns to be accepted internationally, but after freshly found elements matched those predicted by Mendeleev, his patterns could not be denied. In order for the patterns in his "game" to operate, he anticipated that certain elements needed to exist that did not exist at the time. Instead of giving up, he experimented with changing the measured property values to better suit the patterns. He discovered groupings of components with similar features, but he also recognized that there were many exceptions to the forming patterns. Dmitri Mendeleev, the author of the periodic table, began collecting and arranging the known properties of elements in 1869 as if he were playing a game while traveling by train. The contemporary periodic table of chemical elements is as recognizable as a map of the earth, yet it was not always so evident. Read more about the Father of Chemistry and F Block in Periodic Table. Group 1A elements, for example, are soft metals that react aggressively with water.Further elements are classified based on their periodic patterns and characteristics.Elements organized vertically in columns are referred to as "Groups," whereas elements arranged horizontally in rows are referred to as "Periods.".Elements of the Periodic Table are represented by a distinct symbol rather than their full names, as certain elements' names can be lengthy and complex.Elements are organized in increasing atomic number order.Elements are organized horizontally and vertically.The following are some of the characteristics of the periodic table with names. Characteristics of Periodic Table with Names Mendeleev, on the other hand, is awarded sole credit for developing the periodic table. Dimitri Mendeleev proposed his periodic table based on the discoveries of previous scientists such as John Newlands and Antoine-Laurent de Lavoisier. The contemporary periodic table, which we presently use, is a revised and enhanced version of various models proposed by scientists in the nineteenth and twentieth centuries. The first 94 elements of the periodic table occur naturally, but the remaining elements from 95 to 118 have only been synthesized in labs or nuclear reactors. 
As a result, as the atom's energy level rises, so does the number of energy sub-levels per energy level. Elements in the same period, on the other hand, will have an increasing number of valence electrons. Thus, elements in the same group will have comparable chemical characteristics since they have the same valence electron configuration. /GettyImages-1154261034-08fa91cb3d8942c093b9e6b66a26f690.jpg)
Elements are ordered in increasing atomic number order from left to right and top to bottom. They are organized in a tabular format, with a row representing an era and a column representing a group. The periodic table is an organization of all known elements in order of increasing atomic number and repeating chemical characteristics. Dmitri Mendeleev was the first to organize the elements to aid our comprehension. 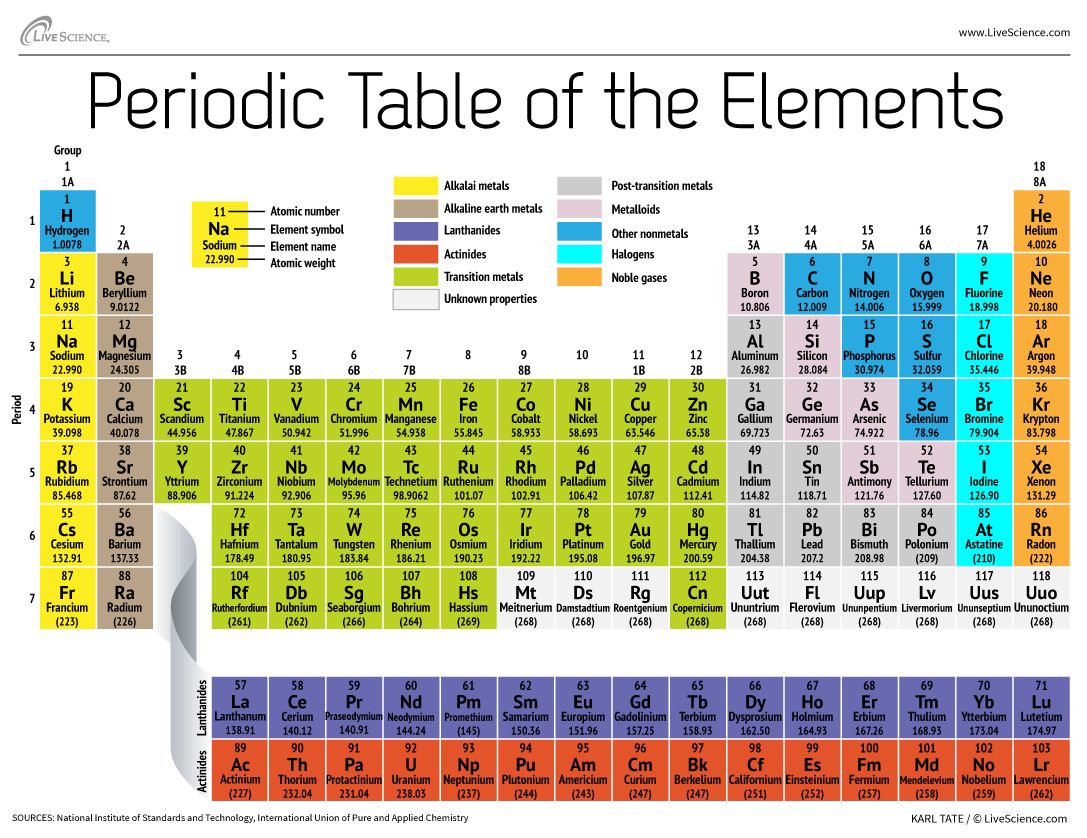
Non-metals are on the left side of the table, whereas metals are on the right. The periodic table (also known as the periodic table of elements ) is organized so that scientists may rapidly determine individual element parameters such as mass, electron number, electron configuration, and distinctive chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
